 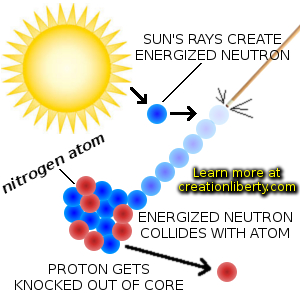 
"voided", shall we say? into the sewer system. Your body does not easily absorb this chemical, so most of the injection is. The dose I was given is about as large as these injections typically get. (Whatever you're being treated for is the greater danger.) The half-life is just long enough for the doctors to have time to take their pictures. Its radiation is extremely low-energy, so the chance of mutation is very low. Either way, I will end up with this equation:īy the way: Technetium-99m is one of the most commonly used radioisotope for these medical purposes. 
Or else I can just deal with the 50% that is left. Since the half-life does not depend on how much I started with, I can either pick an arbitrary beginning amount (such as 100 grams) and then calculate the decay constant after 9.45 minutes, at which point only 50 grams will remain (the other 50 grams will have mutated into some other isotope or element). That is, from t = 0 to t = 9.45, I will have gone from 100% of however much I'd started with (where " 100%" is written as 1, being its decimal equivalent) to 50% of that amount (converted to 0.5 for use inside the formula). I can do this by working from the definition of "half-life": in the given amount of time (in this case, 9.45 minutes), half of the initial amount will be gone. How am I supposed to figure out what the decay constant is? However, I note that there is no beginning or ending amount given. Since the decay rate is given in terms of minutes, then time t will be in minutes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
